 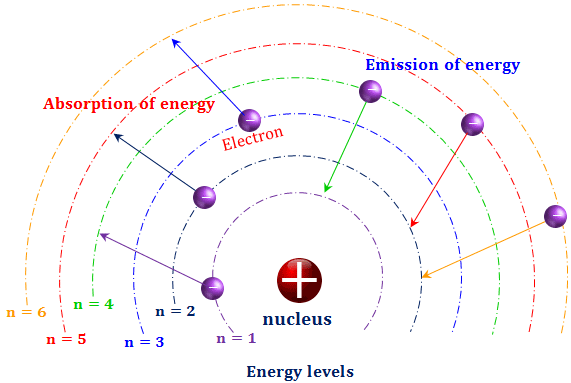
This idea was totally counter-intuitive and required a new physics. The releasing of the photons is responsible for the line spectra given off by elements when excited. Then the electrons would release the photons in quantized amounts to return to ground state. Why?īohr took the ideas of Rutherford (nuclear model), Planck (quanta), Einstein (photoelectric effect and spectroscopy and created the Planetary Model.īohr proposed that electrons were in energy levels (ground state) and absorbed photons of certain frequencies to move to a higher energy level (excited state). The Bohr model (named after Danish physicist Niels Bohr) of an atom has a small, positively charged central nucleus and electrons orbiting in at specific fixed distances from the nucleus. Classical physics says that opposites attract and likes repel, so the negative electrons should be attracted to the positive nucleus. Niels Bohr proposed a model of the atom in which the electron was able to occupy only certain orbits around the nucleus. In 1913, after returning to Copenhagen, he began publishing his theory of the simplest atom, hydrogen, based on the planetary model of. Bohr became convinced of its validity and spent part of 1912 at Rutherford’s laboratory. h E ( 1 n l o w 2 1 n h i g h 2) 13.6 eV. The great Danish physicist Niels Bohr (18851962) made immediate use of Rutherford’s planetary model of the atom. Bohr explained the hydrogen spectrum in terms of electrons absorbing and emitting photons to change energy levels, where the photon energy is. Niels Bohr change the atomic theory by realizing that the electrons did not crash into the nucleus as would be expected in classical physics. Bohrs model calculated the following energies for an electron in the shell, n. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
